Mitochondrial dysfunction, continued, & an ASPEN webinar.
What do medical nutrition specialists know re mitochondria & nutrition? Urolithin A helps promote mitophagy, Niacin therapy & exercise/activity helps. Otherwise, my readers may know more. Review time
This video webinar: The Role of Nutrition Care in Mitochondrial Health - ASPEN 2021, follows up nicely to the last post about mitochondrial dysfunction in cancer. Nature loves a good design; Cancer, POTS, Epigenetics & the One-Carbon Methylation Cycles. (substack.com) Mitochondrial dysfunction is associated with aging, however the speakers in this webinar stress the point that it seems that lack of activity is more associated with the mitochondrial dysfunction than ‘aging’ alone.
// This post is too long for the email, click through. I take you through the some of the slides as screenshots - so lengthy, but not text heavy, and add what Dr. Thomas Seyfried and this video are missing as potential underlying causes of mitochondrial dysfunction, and therefore potential confounding variables in any clinical study with patients. When the experimental and control groups don’t really match, then any results obtained are basically meaningless and the average with be muddied/skewed by the unknown number of people with an underlying causal variable. (Spoiler alert for my astute readers - its the PDK switch and PPAR beta/delta receptors.)
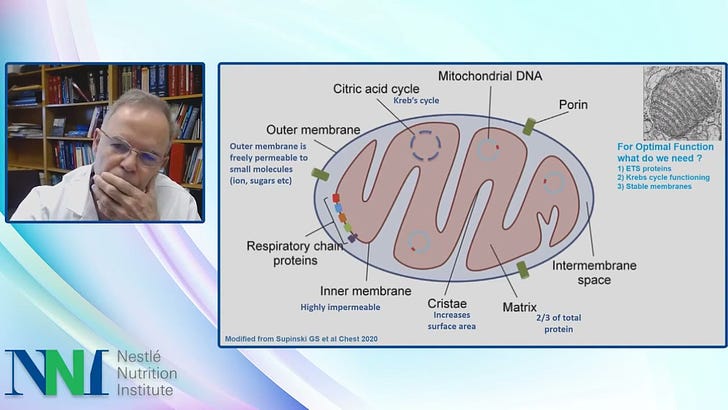
Hosted by Nestle Nutrition Institute and eligible for 1.5 CEU for RDs and DTRs, the webinar was organized by the American Society for Parenteral and Enteral Nutrition (ASPEN). (nutritioncare.org) The speakers include Robert Martindale, MD, PhD, as moderator and first speaker. Bret Goodpastor, PhD is the second speaker and focused on exercise and mitochondria and discussed urolithin A. Eduardo Chini, PhD is the third speaker and focused on NAD+ and niacin or niacinamide therapy for mitochondrial support. He mentioned that chemotherapy has been shown to harm mitochondria.
ICU patients and home-based or residential facility patients on tube feedings are the target group of the speakers, and ASPEN. Nestle Nutrition makes special formula mixtures for special need patient types, and acute care or long-term care. The tone of the speakers is a somewhat focused on ICU patient needs and bed-bound patients. Some people on tube feedings can have mobility too and detach the feeding drip bag, but many others are bed bound and the feeding drips slowly for hours as rapid entry can induce acute problems.
In my professional experience, I read one of ASPEN’s books for an overview of the field. I did apply as a consultant dietitian for a company that provided support and supplies for home-based tube-fed patient and learned more about their business and routine care needs in the process. I don’t remember seeing patients but went through some training with them. It is a very high need population as the mixtures used can have nutrient deficiencies or excesses, or the macronutrient balance can be bad for the condition. Refeeding syndrome is an acute problem with suddenly starting a malnourished person on a full feed drip solution. There are simply risks of aspiration if vomiting occurs and other bolus feeding complications to avoid. Helping renal diet and tube feeding patients are very specialized fields within dietetics and calculating the nutrient needs is important - the dietitian’s math (or doctor’s) will affect the patient’s health - and can kill them if too wrong. Extra training is needed.
The speakers take homes points emphasize that mitochondria need you to get off of the couch and move - or that that is difficult for the bed-bound. I would add, full range stretching movements are important for everyone and assisting the frail patient with some gentle daily exercises is needed. Stronger bed-bound patients can do arm exercises maybe - conduct along with some energetic classical music. Band conductors have been found to live longer lives than average. Get those arms above the head if possible.
Point they did not emphasize well but was briefly mentioned and then discussed later without reaching the answer - very strenuous exercise is a risk for mitochondrial dysfunction - and can reduce immune function temporarily. The answer - polyphenols as part of the diet or a supplement prior to or shortly after the exercise can help reduce the oxidative damage and shorten the drop in immune function. The strenuous exercise creates myokines which are like cytokines or adipose tissue’s leptin, an adipokine/adipocytokine, and add to an inflammatory state. (Nieman, 2020) Without extra antioxidant support the oxidative stress level would lead to mitochondrial damage.
Reference - included in a virtual conference about the microbiome which I made a document of all the links and poster sessions (IECN 2020 document) See the Keynote session: Professor David Nieman, A Multi-Omics Approach to Interpreting the Influence of Polyphenols in Countering Exercise-Induced Physiological Stress.
Doesn’t that pomegranate look delicious, and the blueberries?
The nutrients that may or may not help mitochondria were discussed briefly by the first speaker, the slide below is the second speaker. The first mentioned nutrients that have been studied singly regarding mitochondria improvement, and most were not found helpful singly, except for melatonin, the cofactor acetyl-carnitine and the antioxidant alpha lipoic acid.
See the Resources section at the end if you are just interested in What to take? Cautions, other guidance. Mitochondrial support nutrients are a topic that I have made guidance graphics for. What to take? That may depend on how sick you are. Adding a few or one thing at a time can help to watch for any negatives. Otherwise, it is good to continue for a few months for optimal healing time. I do take quite a few/most of the lists, but not quite all. I consider the Bs, CoQ10, and alpha lipoic acid to be near the top of my shorter list. Omega 3 fatty acids EPA/DHA, adequate protein, and Nrf2 promoters. Epsom salt soaks for topical bypassing any GI malabsorption, and a good trace mineral mixed supplement.








The first speaker provided an overview of research regarding nutrient supplementation for mitochondrial support and mentioned them all singly. He had a summary point that maybe a mixture of the support nutrients are needed, but the best mix is not known. It would vary somewhat with the patient’s individual issues. In general though, I think it is quite safe to assume that problems in the “one-carbon methylation” cycles, may involve a lack of methylation of folate - so any study that looks at how helpful cyanocobalamin or folic acid is to the patient - is a big waste of time in my opinion and a little silly too. Or a lot. Why would you give unmethylated supplements to someone who may have a methylation problem? And any ICU patient should be assumed to be in metabolic distress and doesn’t need to be wasting any effort on unnecessary chemical steps.
It takes ~ 14 chemical steps to make one nucleotide and one mRNA may need 100s of nucleotides to be created. No protein is going to be created until first the matching mRNA was created. Nutritional Yeast Flakes are a vegan source of nucleotides. People who use animal products probably get some, but vegans may be quite low unless they use Nutritional Yeast Flakes (about 2 teaspoons per day may be adequate). I buy an unfortified brand because it tends to be fortified with folic acid and cyanocobalamin. Using those is bad for people who can’t remethylate well because it fills the body with a not quite right chemical that may interfere with the body’s access to any real folate or methyl-cobalamin that was eaten in the diet.
The other big missing fact/facts that the Cancer video and this video didn’t include is the potentially confounding variables of a high fat/high saturated fat diet and/or Retinoid Toxicity. Either may be a direct and ongoing cause of mitochondrial dysfunction - the PDK switch away from the energy efficient and less waste producing Citric Acid Cycle to the less efficient fermentation of glucose or glutamate which produces lactic acid in excess adding to oxidative stress in the mitochondria and eventually the body if exertion continues.
Diet with moderate to low saturated fats. A confounding variable to any study on mitochondrial function - a high saturated fat diet is the normal cause for the PPAR beta/delta inhibition of PDK and the Citric Acid Cycle by limiting CoA production . . . which blocks the Citric Acid Cycle from functioning, and the mitochondria then switch to using fat for energy. (Tyagi, et al, 2011) (Zhang S, et al, 2014, Fig. 2)
This PDK switch is a standard part of metabolism and would be lifesaving when food was scarce - a rapid switch to using whatever type of energy was most available at the time. In modern life saturated fats can be a significant part of the diet. Using fermentation for energy production is more inefficient for energy needs though, and produces more waste and lactate build up from the accumulating pyruvate. Elevated pyruvate levels are seen in ME/CFS. (Anderson and Maes, 2020)
What is a high fat diet? In humans 20-30% calories from fat are typical, with a goal of 10% or less to be from saturated fats and zero from trans fats. Above 35-50% of the total calories from fats is considered high fat for human meal planning. Saturated fats in particular trigger PPAR inhibition of CoA production for Citric Acid Cycle use by mitochondria – they are switched to fermentation of fats instead – meant to be temporary for survival, becomes mitochondrial dysfunction with many modern diets.
Lack of nutrients and cofactors needed in the Citric Acid Cycle can be involved and a high saturated fat diet may be adding to the shift in mitochondrial energy production to fermentation instead of using the more productive and less waste producing Citric Acid Cycle (pyruvate is a waste product of fermentation).
The curve ball is that Retinoid Toxicity may also be a confounding variable causing a chronic inhibition of PDK by PPAR activation by 9-cis-Retinoic Acid - leading to mitochondrial dysfunction - ongoing use of fermentation instead of the Citric Acid Cycle.
The ability to use the Citric Acid Cycle can be turned off by 9 cis-Retinoic acid activated PPAR beta/delta receptors, (Reay and Cairns, 2020), inhibiting PDK which then prevents production of Acetyl CoA, which is needed for the cycle to occur within mitochondria. (Tyagi, et al, 2011)
Patients may have both an underlying over-activation of retinoids, and be eating too high fat or high saturated fat diet. (Butter, lard, shortening, some margarine, cheese, some meats and dairy products more than others, coconut oil or products, palm oil. Vegans do benefit from including some coconut oil in their diet as a cholesterol precursor as the vegan diet has no cholesterol and we do need some to help reduce inflammation and have adequate health. It got demonized unfairly along with fats by the sugar industry about 50-60 years ago with fraudulent research claims about fats and heart disease to turn attention away from the risk of carbohydrates for vascular health.
Even in my younger days, I turn bright red when exercising and was not hired as an aerobics instructor because I made the routines look too strenuous - I looked hot and bothered rather than calm, cool, and in command of a class of exercisers looking for a role model to follow. That is also a reminder of the take home point from the last post - do not get overheated. That is doubly important for me. One of my gene alleles is affected negatively by higher temperatures - more dysfunctional if I am above 98.6’C or so.
Histamine excess or the niacin flush can also cause skin reddening at times.
Resources

See https://www.jenniferdepew.com pages Nutrients and page Cofactors for links to more information and a basic overview.
Disclaimer: This information is being shared for educational purposes within the guidelines of Fair Use and is not intended to provide individual health guidance.
Reference List
(Anderson and Maes, 2020) Anderson, G., Maes, M., (2020). Mitochondria and immunity in chronic fatigue syndrome, Progress in Neuro-Psychopharmacology and Biological Psychiatry, 103, 109976, ISSN 0278-5846, https://doi.org/10.1016/j.pnpbp.2020.109976. https://www.sciencedirect.com/science/article/pii/S027858462030292X Full text pdf: Available at: https://www.dropbox.com/s/vofaipxk1tknxu0/1-s2.0-S027858462030292X-main.pdf?dl=0 (Accessed: 21 October 2022)
(ASPEN/nutrition and mitochondria) The Role of Nutrition Care in Mitochondrial Health - ASPEN 2021, youtube.com, https://www.youtube.com/watch?v=kpm0dqeuXmA
(Nieman, 2020) Nieman, D. (2020). A Multi-Omics Approach to Interpreting the Influence of Polyphenols in Countering Exercise-Induced Physiological Stress, in Proceedings of the 1st International Electronic Conference on Nutrients - Nutritional and Microbiota Effects on Chronic Disease, 2–15 November 2020, MDPI: Basel, Switzerland, doi:10.3390/IECN2020-06978 https://sciforum.net/paper/view/conference/6978
(Reay and Cairns, 2020) Reay, W.R., and Cairns, M.J. (2020). ‘The role of the retinoids in schizophrenia: genomic and clinical perspectives’. Mol Psychiatry 25:706–718. https://doi.org/10.1038/s41380-019-0566-2 Available at: https://www.nature.com/articles/s41380-019-0566-2 (Accessed: 21 October 2022)
(Tyagi, et al, 2011) Tyagi, S., Gupta, P., Saini, A.S., Kaushal, C., Sharma, S., (2011). The peroxisome proliferator-activated receptor: A family of nuclear receptors role in various diseases. J Adv Pharm Technol Res. Oct;2(4):236-40. doi: 10.4103/2231-4040.90879. PMID: 22247890; PMCID: PMC3255347. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3255347/ (Accessed: 21 October 2022)
(Zhang S, et al, 2014, Fig. 2) Zhang, S., Hulver, M.W., McMillan, R.P. et al., The pivotal role of pyruvate dehydrogenase kinases in metabolic flexibility. Nutr Metab (Lond) 11, 10 (2014). https://doi.org/10.1186/1743-7075-11-10 Available at: https://nutritionandmetabolism.biomedcentral.com/articles/10.1186/1743-7075-11-10 Figure 1: https://nutritionandmetabolism.biomedcentral.com/articles/10.1186/1743-7075-11-10/figures/1 Figure 2: https://nutritionandmetabolism.biomedcentral.com/articles/10.1186/1743-7075-11-10/figures/2 (Accessed: 26 Oct 2022)














Amazingly informative article. I have yet to check out the links and resources. Thank you for including them!